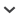How the carbon balance of arctic ecosystems responds to climate warming will depend on the changes in carbon assimilation capacity of tundra plant species. Along with air and soil warming, one of the consequences of warming likely to be important for carbon assimilation of tundra plant species is an expected 40% increase in growing season length. We examined the effects of a lengthened growing season and soil warming on the photosynthetic capacity of seven tundra plant species from four growth forms that comprise >90% of the vascular cover of wet tussock tundra. Maximum photosynthetic capacity of these key species was relatively unchanged by the manipulation that significantly altered growing season length, active layer depth, and soil temperatures. Highest photosynthetic rates were found for the forb, Polygonum bistorta, and the lowest for dwarf evergreen shrubs. Seasonal patterns revealed that plants maintained relatively high light-saturated photosynthetic capacity (Amax) values throughout most of the growing season. Interannual variation was significant, but differences were small for most species. The study shows that tundra species operate within a relatively narrow range for maximum photosynthetic capacity with this maximum seldom being reached under ambient conditions. Thus, when evaluating the effects of climate change on tundra ecosystem carbon uptake, species composition and total photosynthetic leaf area should be considered first. These two factors will affect the system carbon exchange capacity during climate warming more so than species-level assimilation capacity.
Introduction
During the past two decades, evidence has accumulated that indicates an alarming rate of warming in Arctic Alaska (Chapin et al., 1995; Oechel et al., 1995; IPCC, 2001, 2007; ACIA 2005; Camill, 2005; Hinzman et al., 2005). These increasing temperatures can likely be attributed to anthropogenic practices that continue to increase atmospheric concentrations of carbon dioxide (Maxwell, 1992; Keeling et al., 1995). This warming is having a considerable effect on the physical environment that drives weather patterns at northern latitudes, such as a thinning of the pack ice in the Western Arctic Ocean (Maxwell, 1996; Maslanik et al., 1999). Reduction in the sea ice has increased surface water movement and ocean mixing, which has a direct effect on the region's weather patterns, primarily precipitation events (Carmack and Kulikov, 1998).
The combination of changing weather patterns and increasing winter temperatures has led to predictions that the arctic growing season may increase by as much as 40% by the middle of this century (Maxwell, 1992, 1996). This increase can be attributed to an earlier snowmelt during the spring and a later accumulation of snow during the fall (Maxwell, 1992). An increase of such large magnitude will have a significant effect on many environmental factors important for maintaining plant physiological activity, community structure, and ecosystem function (Harte et al., 1995). Among these factors is permafrost, which has controlled the structure and function of arctic plant communities over tens of thousands of years (Billings, 1996). Permafrost limits plant development through the restriction of root growth and root kinetics in the active layer, which is just slightly above freezing during the course of the growing season (Marion and Kummerow, 1990). These low soil temperatures have been shown to restrict the ability of roots to move water and nutrients needed for shoot physiological activity (Starr et al., 2004). The permafrost of Alaska is warming, thus causing an increase in the active layer (Lachenbruch and Marshall, 1986; Maxwell, 1992; Osterkamp and Romanovsky, 1997; Camill, 2005). Changes in soil conditions can affect nutrient cycling, soil moisture content, and soil aeration (Lachenbruch and Marshall, 1986; Manabe and Wetherald, 1986; Kane et al., 1992), leading to dramatic changes in the physiological status of arctic plant communities (Shaver et al., 1992; Hobbie and Chapin, 1998).
A lengthened growing season can also lead to water stress and a decrease in transpiration when depth of thaw increases are coupled with periods of warm-dry weather (Starr et al., 2000). This decrease in water within the rhizosphere can lead to a reduction in plant growth (Oberbauer and Miller, 1982). If water stress is prolonged, photosynthetic activity will diminish (Kramer and Boyer, 1995). At the ecosystem level, this can reduce the carbon sink capacity (McKane et al., 1997; Oechel et al., 1998).
If water is not limiting, physiological activity may increase in response to warmer temperatures. Studies in arctic tundra have shown that dominant species, such as Eriophorum vaginatum, increase growth as a result of increased ability to acquire nutrients as root kinetics improve with warmer soil temperatures (Kummerow and Ellis, 1992). The deciduous shrub Salix pulchra increased the diameter of xylem vessels in response to warmer temperatures, leading to increased physiological activity compared with plants grown at ambient temperatures (Gorsuch and Oberbauer, 2002). If this pattern of physiological activity holds within all significant growth forms across the Alaskan tundra, then the carbon sink capacity of the region could increase.
Little is known however, about the maximum physiological capacity of tundra plants under natural conditions or in relation to changing season length and soil temperature. Most studies of the photosynthetic capacity of arctic species under natural conditions have been during short periods at peak season when weather was most favorable, mid June through mid August (Oberbauer and Oechel, 1989; Wookey et al., 1994; Oberbauer et al., 1996). The need to develop a comprehensive understanding of the physiological capacity of tundra plants over the course of their entire physiological period and under conditions related to climate change sets the foundation for this study.
The objective of this research was to study the seasonal patterns in seven vascular plant species to changes in the growing season length and increased soil temperatures. The study species included members of four growth forms—graminoids, perennial forbs, and evergreen and deciduous dwarf shrubs. The species were as follows: Eriophorum vaginatum L. and Carex bigelowii Torr. (graminoids), Ledum palustre L. and Vaccinium vitis-idaea L. (dwarf evergreen shrubs), Betula nana L. and Salix pulchra Cham. (dwarf deciduous shrubs), and Polygonum bistorta L. (forb). These seven vascular species comprise >90% of the vascular ground cover of the Alaskan moist acidic tussock tundra and play a key role in the ecosystem physiology (McKane et al., 1997).
We hypothesized that deciduous shrubs and graminoids would have the greatest physiological plasticity, thus being able to take advantage of a longer growing period. In related climate change experiments, these growth forms have come to dominate tundra communities (Chapin et al., 1995; Walker et al., 2006) suggesting they have phenological plasticity that allows them to take advantage of changing conditions related to climate warming. The evergreens and forbs were predicted to have a more limited range of physiological capacity and adjustment to manipulations. For the evergreens, this response should result from their strategy of maintaining a low physiological activity annually, but persisting over a long periods. For the forb P. bistorta, we hypothesized that its fixed phenological pattern would limit its ability to adjust physiological capacity (Starr et al., 2000). In testing these hypotheses, our aim was to develop a comprehensive understanding of the potential CO2 assimilation rates for these species in relation to environmental conditions and predicted climate changes.
The objectives and hypotheses of this study were tested using a series of field manipulations in which the growing season was extended by removing the snow cover and maintaining plots snow free in the early spring and preventing snow accumulation and hard frost in the early fall. In addition, a treatment was included in which the soil of a group of extended season plots was warmed using greenhouse heating wires to examine the effects of increased soil temperatures.
Methods
Study Site and Design
This experimental manipulation was conducted in acidic tussock-dwarf-shrub tundra, approximately 1.5 km southwest of Toolik Field Station, Arctic Alaska (68°38′N, 149°34′W, elevation 730 m) as part of the International Tundra Experiment (ITEX) (Molau and Mølgaard, 1996; Henry and Molau, 1997). Eriophorum vaginatum tussocks dominate this community, with intertussock spaces filled with a combination of dwarf deciduous and evergreen shrubs and peat-forming mosses. A detailed description of this community can be found in Bliss and Matveyeva (1992).
This study was conducted during years 3 to 5 (1997–1999) as part of a long-term manipulation of the growing season length and soil warming. The study consisted of 18 plots (each 1.5 × 1.5 m) in a complete randomized block design with 6 blocks and 3 treatments—extended season (ES), extended season + soil warming (ESW), and control (C). These primary study plots were reserved exclusively for nondestructive measurements, including the photosynthesis measurements in this study. Four additional plots of each treatment were established to provide treatments for tissue nutrient sampling. The soil warming experiment was initiated by placing cold frame greenhouse heating wires approximately 10 cm below ground surface in 1994, one year prior to the start of the field manipulations. The number of replicates and size of the plots for the experiment was limited by the intense labor needed to remove the snow quickly at the start of the growing season and to keep plots snow free.
For all soil warming plots, a 1400-watt generator was used to power the greenhouse heating wires 2 h daily, around solar noon, for a 0.4 MJ m−2 d−1 of energy input to the soil (equivalent to 20–30% of the daily soil heat flux for this vegetation during summer months [Eugster et al., 2005]). This magnitude of increase in the energy of the soil is similar to that predicted with global warming (Maxwell, 1992). During the 1997 and 1999 field seasons, soil warming was initiated on 3 May. In 1998 the soil warming was initiated on 29 April.
The growing season was lengthened by carefully removing snow at the beginning of the season. We define the growing season as the period between the day our control plots became 90% snow free to 4 September, when our seasonal manipulation was discontinued. Care was taken not to damage the canopy or remove any of the litter in the plots. In 1997 and 1999 snow removal started on 1 May and was completed by 3 May. In 1998 the manipulation was initiated on 27 April and snow removal was completed by 28 April. The early initiation of the manipulation in spring of 1998 was in response to reports of a very low snow load on the North Slope of Alaska. This insured that some period of early season manipulation would take place. Snow depth at the site prior to removal ranged between 48 and 80 cm, 35 and 70 cm, and 38 and 71 cm for 1997, 1998, and 1999, respectively. Once the snow load was removed, A-frame tents made of greenhouse plastic were placed on the plots. These tents increased the air temperatures approximately 3.0°C, resulting in rapid melt of the remaining snow. The ends of the A-frames were opened except during snow or windstorms so as to minimize increases in air temperatures on the plots. The intention of our study was to lengthen the growing season with little manipulation of air temperature.
The A-frames remained on the plots until the threat of early season frost damage or snowstorms no longer existed (approximately 1 June). For late season extension, the A-frames were placed on the plots 14 August each of the three seasons. This technique reduced late-season frost damage and left the plots snow free through 4 September, when the treatments were ended for the season.
Physiological Measurements
Maximum light-saturated photosynthetic capacity (Amax) and stomatal conductance of one randomly selected individual of the study species on each primary plot was measured using a Li-6200 portable photosynthesis system with a 0.25 chamber (LI-COR Inc., Lincoln, NE) on a weekly basis, weather permitting. Monofilament line strung across the upper and lower chamber openings insured that the leaves of the different species were oriented perpendicular to the light source. In the case of Ledum palustre shoots, which consist of a tight bundle of whorled leaves, shoot tips were placed in the chamber in orientations that minimized shading of leaves within the whorl. To obtain an Amax measurement, the Li-6200 was equipped with a Quantum Devices Q-beam 2001 red spectrum light source set at a photosynthetic photon flux density (PPFD) of 1500 µmol m−2 s−1 (Quantum Devices Inc., Barneveld, WI). This light level had been shown to be above the light saturation point for the individual species of this study and has been used in the past to determine Amax (Oberbauer and Oechel, 1989). Each plant was allowed 5 min to acclimate to the light conditions prior to taking the measurement. The photosynthesis system was calibrated daily before each use with a primary standard (1%) of CO2 in air (Scott-Marrin, Inc, Riverside, CA) or a secondary standard calibrated using the primary standard.
All measurements were expressed on a per unit area basis. Because the experimental manipulation was a long-term study, leaf samples used in photosynthesis measurements could not be removed from the primary study plots for determination of leaf area or dry weight. Leaf areas of P. bistorta were determined using leaf tracings measured with a LI-3050A/4 leaf area meter (LI-COR Inc., Lincoln, NE). To determine leaf areas for the other species, a regression model between maximum leaf length and/or width and leaf area was developed for each species.
Microclimate Measurements and Soil Properties
Weather data were acquired from the micrometeorological station of the Long-Term Ecological Research Project (LTER) terrestrial site located southwest of Toolik Field Station approximately 300 m north of our manipulation. Average daily air temperatures and precipitation were recorded for each day of the three field seasons. All weather data reported are based on the time frame of our seasonal experiment, 26 April to 14 September. In addition, soil temperature was measured at 5 cm. in each of the treatment plots using copper-constantan thermocouples with a Campbell 21X datalogger and AM25T solid state multiplexer (Campbell Scientific Inc, Logan, UT). Measurements were taken every minute and saved hourly. Soil moisture was measured in five of the study blocks using Campbell CS615 water content reflectometers (Campbell Scientific Inc, Logan, UT) placed at 45° with respect to the soil surface, thereby integrating soil moisture across the top 20 cm of soil. These sensors were installed during the 1997 field campaign once depth of thaw surpassed 20 cm, and the first readings were on 3 July. The CS615 has not been calibrated for the soils at this site, so the reported values should be considered relative estimates of volumetric water content. These measurements were taken twice weekly with a handheld reader (O'Brien and Oberbauer, 2001).
Standard thaw probing techniques were used weekly to determine soil active layer depth over the course of the study (Oberbauer et al., 1991) in both the primary and the nutrient sampling plots. Measurements were taken at two points in intertussock spaces that were randomly chosen in each plot at the beginning of each field season.
Statistical Analysis
For statistical analysis, the individual plant was treated as the replicate. The main effects of species, treatment, time of season, and their interactions were analyzed for each study year using a split-plot design with PROC GLM in SAS 9.1 (SAS Institute, Inc, Cary NC). The effects of treatment on individual species were analyzed using Repeated Measures ANOVA in the statistical software package Superanova (Abacus Concepts, Inc., Berkeley, CA). Data sets were transformed prior to repeated measures test using the most appropriate method based on the skewness of the data (Zar, 1984; Stevens, 1996). Once all data sets were normally distributed they were tested for homoscedasticity with the Bartlett χ2 test using Statistica (StatsSoft, Inc., Tulsa, OK). If significance between the treatments was seen with Repeated Measure ANOVA, a Bonferroni-Dunn test was then used to determine differences among the treatments.
In cases where data could not be normalized, nonparametric Kruskal-Wallis analysis of variance (ANOVA) was used to test for interactions between the blocks, treatments, and plots within and among the years of study. In all cases, block had no effect in the study. Based on these findings, the results reported are the differences found among the treatments during each individual year of study and the differences seen among the three years. For comparisons among years, the range of dates of the year with the shortest sample season was used to calculate seasonal mean Amax. In cases where differences were seen among the treatments, a Mann-Whitney U test was then used to determine which treatments differed. All nonparametric analyses were done using the statistical software package Statistica.
To determine what environmental parameters accounted for the variance seen in the photosynthetic data, a Principal Component Analysis (PCA) was first run for each species to determine the covariance of environmental variables that may have affected photosynthetic activity. Following PCA, multiple regressions were run on the PCA factors and independent environmental variables. PCA and multiple regressions were preformed using Statistica.
Results
Microclimate Properties
Mean daily air temperatures 1.0 m above the surface were 7.1 ± 5.8°C SE for 1997 with the maximum temperature of 16.0°C on 4 August and the minimum temperature of −11.1°C on 9 May. In 1998, the average temperature was 6.4 ± 7.7°C SE with the maximum of 18.7°C, occurring on 21 July and the minimum temperature of −16.8°C on 4 May. During the 1999 field season temperatures were slightly cooler than the previous two years with the average of 5.6 ± 8.9°C SE, maximum of 18.9°C occurring on 5 August and the minimum of −19.2°C occurring on 16 May.
Experimental manipulations resulted in approximately a 30% extension in the normal growing season length. The variability of spring weather conditions directly affected the length of the early season manipulation. Over the course of the three years of study, manipulated plots became snow free on average by 7 May, 1 May, and 7 May for 1997, 1998, and 1999 respectively. The control plots became snow free on 4 June, 19 May, and 23 May. These extensions coupled with the late season extension from 14 August to 4 September accounted for a 49, 40, and 37 d extension of the growing season for each year, respectively.
A general pattern was found in the maximum depth of thaw within the plots; in all cases the ES plots had the greatest depth of thaw, followed by that of the ESW plots and the controls. These differences however, were not statistically significant (Table 1). In addition, depth of thaw did not differ significantly across the years of study (Table 1).
Table 1
Effects of seasonal manipulation on maximum depth of thaw, seasonal average soil temperature (5 cm depth), and seasonal average volumetric water content of the soils (top 20 cm integrated) during the three field seasons. Depth of thaw values in same rows are mean depth (cm) for 26 August of each year. Soil temperature is the average temperature for the period 18 May–2 September. Soil moisture is the average volumetric water content for the growing season (n = 5). Letters representing differences among the treatments are based on Mann-Whitney U-test. P-values represent results from Kruskal-Wallis test among treatments and years.

Soil temperatures at 5 cm depth were generally lowest on the controls, followed by the ES plots, with the ESW plots having the highest temperatures (Table 1). However, these differences were not statistically significant across the treatments or years (Table 1).
Volumetric water in the upper 20 cm of the active layer differed significantly among the treatments in two years of the study (Table 1). In 1998 and 1999, the manipulation plots, ES and ESW, were similar, but differed significantly from the controls (Table 1). However in 1997 for which we had data for only part of the growing season, no differences among the individual treatments were found.
Physiological Measurements
Comparisons of the main of effects of species, treatment, and time of season (weeks) on photosynthesis showed three significant effects for all three years of the study, the effects of species, weeks, and the species*week interaction (Table 2). These results indicate that species differed in photosynthesis capacity, photosynthetic capacity significantly varied over the season, and the seasonal pattern of photosynthesis over the season differed among species. Differences in species Amax were related to growth form. The deciduous shrubs had the highest photosynthetic capacity while the evergreen shrubs had the lowest (Fig. 1). The evergreens and semi-evergreen species, L. palustre, V. vitis-idaea, and E. vaginatum, had the longest periods of photosynthetic activity during the study, while P. bistorta had the shortest, with the other three species intermediate (Fig. 1). The main effect of treatment was close to significance in 1997 and 1998 but not in 1999. The treatment*species interaction was close to significance in 1997.
Table 2
Results of split-plot ANOVA testing main effects of species, time of season (weeks), and interactions for maximum photosynthetic capacity for 1997–1999. ** highlight P < 0.01.

Figure 1
Seasonal photosynthetic capacity (Amax) of seven species over the course of the study, 1997–1999. (A) Eriophorum vaginatum, (B) Carex bigellowii, (C) Betula nana, (D) Salix pulchra, (E) Vaccinium vitis-idaea, (F) Ledum palustre, and (G) Polygonum bistorta. Values represent means ±SE (n = 6).

Stomatal conductance was subjected to the same analysis above. The main effect of week was significant in 1998, and significant species*week interactions were found in 1999 (data not shown). Similar to photosynthesis, the effect of treatment was close to significance in 1997 (P = 0.061).
Few statistically significant effects of treatment on Amax were found for the seven species when tested for differences within the individual species and years (Fig. 1, Table 3). Significant effects were present in 1997 for V. vitis-idaea and in 1999 for P. bistorta, but post-hoc comparisons with a Bonferroni-Dunn test failed to show differences among treatment means. Significant differences in Amax for weeks in the growing season were found for all species in all years except for V. vitis-idaea in 1999 (P = 0.0848, Table 3). In four of a possible 21 cases, significant treatment by time of year interactions were found, suggesting differences in the temporal patterns in response to treatment in these cases (Table 3). Two of the four cases were Polygonum bistorta (1997, 1999) in addition to E. vaginatum in 1997 and C. bigellowii in 1998.
Table 3
Effect of seasonal manipulations on leaf Amax (µmol CO2 m−2 s−1) for the seven study species using Repeated Measures ANOVA for 1997, 1998, and 1999. ** highlight P < 0.01, * highlight P < 0.05.

All species except B. nana had statistically significant differences in Amax between at least two of the seasons (Table 4). In all growth forms except the deciduous shrubs, photosynthetic capacity decreased in 1999. The deciduous shrubs, in contrast, increased photosynthetic capacity that year (Table 4).
Table 4
Effect of study year on Amax (µmol CO2 m−2 s−1). Values in rows are seasonal means of Amax, different letters indicate a significant difference among the years based on Mann-Whitney U-test. P-values represent results from a Kruskal-Wallis test among the years of the study.

To further explore the basis for the time of season effect, principal component analyses (PCA) were used to simplify the environmental and physiological factors that might be important for seasonal differences. PCA produced two significant factors among environment variables measured during the study. Factor 1 condensed air and leaf temperature into one variable, while Factor 2 included year and relative humidity. Multiple regression analysis of the environmental variables showed species specific relationships among photosynthesis and environmental variables. Factor 1 and CO2 concentration accounted for 20% of the variance in the Amax of Eriophorum vaginatum (P < 0.0001), while day of year and CO2 concentration accounted for 16% of the variation in Amax for Carex bigellowii (P < 0.0001). For Betula nana, 18% of the variability in Amax could be accounted for by Factor 2, CO2 concentration, intercellular concentrations of CO2, and stomatal conductance, P < 0.0001. The other deciduous shrub, Salix pulchra, showed no statistical significance between the tested environmental variables and Amax, P = 0.636. Only 4.5% of Amax variance could be accounted for in Ledum palustre, by Factor 2 and CO2 concentration. Again with the evergreen, Vaccinium vitis-idaea, a minimal amount of the variance could be accounted for by day of year, relative humidity, and the individual year of study, R2 = 0.08 and P < 0.0001. For P. bistorta, 12% of the variance in Amax could be accounted for by the day of year, P < 0.0001.
Discussion
We hypothesized that deciduous shrubs and graminoids would have the greatest physiological plasticity and thus be able to take advantage of the conditions established by the manipulation of the growing season, either in the form of greater Amax or a longer period of active photosynthesis. Instead, no significant trends were found to support greater plasticity of deciduous shrubs and graminoids. The maximum photosynthetic capacity of all four growth forms was limited to a relatively narrow range when compared across the treatments. Only two species showed any significant treatment effects, each in only one of the three study years.
One interpretation of these results is that the lack of change in physiological capacity implies that vascular tundra plants have internal constraints that limit their ability to alter the maximum physiological processes in response to manipulations related to climate warming (Hobbie and Chapin, 1998; Starr et al., 2000). This idea is strengthened by the relatively modest differences in seasonal mean Amax among the three years in this study and by prior research that found similar Amax values from a site with cooler weather conditions and a shorter growing season (Oberbauer and Oechel, 1989).
An alternative view is that the lack of significant differences implies strong homeostatic regulation in response to snow removal treatments given that despite these environmental changes, plants maintained similar photosynthetic capacities over the growing season. The one exception to this lack of response to the manipulation was seen in the Amax measurement of P. bistorta. Evidence suggests that this forb has a fixed phenological pattern that limits its ability to adjust to environmental changes (Walker et al., 1995; Diggle, 1997; Chasan, 1998; Starr et al., 2000). Polygonum bistorta leafed out earlier in response to the treatments but also senesced earlier, with a fixed growing season length (although leaves that would normally develop the following year sometimes emerged in late July and August on the treatment plots). This fixed growing season length appeared as a significant weeks*treatment interaction for P. bistorta in two of the three study years.
This weak response is consistent with studies that suggest that plants at low arctic sites such as Toolik are less sensitive to manipulative changes than those at higher latitude sites (Jones et al., 1997; Arft et al., 1999), because the Low Arctic experiences a larger variation in natural weather conditions over the course of the growing season. The wide range of physiological tolerances for low arctic plants (McGraw, 1987) may explain the lack of correlation between the physiological measurements and the manipulations.
Another interpretation is that the experimental manipulation did not sufficiently affect the environment to alter the physiological capacity of the plants. The results of Arft et al. (1999), Hollister and Webber (2000), and Walker et al. (2006), however, imply that small temperature differences, even air temperature differences less than 2°C, can have significant physiological effects. Chapin and Shaver (1996) found that photosynthetic capacity of tundra plants increased in response to mild greenhouse warming. In our study, the growing season was lengthened approximately 30%, much of which was in the critical early growing season. The removal of snow early in the season increases solar inputs, increasing seasonal average soil temperatures at 5 cm depth approximately 1°C, and with heating cables soil temperature increased approximately 2.0°C (Oberbauer et al., 1998). Increased soil temperature had a direct effect on the maximum depth of thaw across the manipulation plots. Although seasonal maximum depth of thaw did not differ significantly among the treatments, in part as a result of the relatively small sample size, thaw depth on the snow removal treatments was greater in early season than in the control plots. Interestingly, the ES plots had slightly greater depth of thaw than the ESW plots, a trend also found in prior study years (Starr et al., 2000). This could be the result of chance microtopographic variation or lateral displacement of the heat from the heating wires as a consequence of reduced surface soil moisture in the ESW plots (Table 1). Regardless, timing of early season thaw may be more important than seasonal maximum because irradiance for photosynthesis is maximal and most of the growth occurs early in the season.
Changes in soil temperature and thaw depth may have both direct and indirect effects on the photosynthetic capacity of tundra plants. Direct effects include release from photosynthetic limitations through increased stomatal conductance by improved root/water status. Indirect effects include improved nutrient availability through increased mineralization rates and greater exploitable soil volume with greater depth of thaw (Shaver et al., 1991). In a related growth-chamber study Starr et al. (2004) found a direct positive relationship between soil temperature and stomatal conductance and Amax of the two dominant sedges measured within this study, but differences of 5°C were needed to show significant photosynthetic differences. Tussock tundra is known to be strongly nutrient limited (Chapin and Shaver, 1985). In nitrogen-limited environments, such as tussock tundra, the costs to increase leaf area or photosynthetic apparatus are great and thus plants may limit their growth or physiological capacity (Kudo, 1999). If increases in nutrient availability did occur, the species would likely allocate these additional resources to increased productivity (Jonasson and Shaver, 1999). Bowman et al. (1995) showed that an increase in nutrient availability within the alpine tundra caused an increase in photosynthetic capacity during the first year when the number of leaves had been predetermined in the previous year, but in following years the additional nutrients were allocated toward increased total leaf area without an increase in photosynthetic capacity. Our study included years three to five of these manipulations, and treatment plants should have been adjusted to any higher nutrient levels. Indeed, in all years of the treatment leaf area was greater on the treatment plots than controls (Oberbauer et al., 1998, unpublished data).
Effects of early snow removal are not all physiologically beneficial. Snow-free plants may be exposed to damaging low temperatures early in the season that snow covered plants are not (Inouye, 2000). The effects of mid-season frosts on plants that have developed under warmer temperature regimes are likely to be more severe (Inouye et al., 2002; Gorsuch and Oberbauer, 2002). The loss of evergreens on the snow removal plots supports this idea (Oberbauer, unpublished data). Thus, these treatments reflect the balance between the positive and negative effects of early snow removal on photosynthesis.
A further complication was that despite the early emergence and photosynthetic activation of plants on the treatment plots, because the controls were still under snow and could not be measured without disrupting the experiment, we could only statistically test with repeated measures sample periods when all three treatments were snow free. However, leaves were photosynthetically active on the treatment plots in early season for all species whenever tested. Totalized Amax using all measurement dates within a season, similar to the seasonal means over the snowfree dates, did not significantly differ among treatments for most species. Only Salix pulchra and Vaccinium vitis-idaea differed significantly in 1997 among treatments for totalized Amax, when testing all species among the years of the study. The pattern of Amax over the course of the growing season was that generally seen for vascular plants within the tundra and alpine regions; immediately following snow melt, photosynthesis increases to its maximum capacity to take advantage of the short growing season. Towards the end of the growing season, a retranslocation of nitrogen from photosynthetic apparatus to storage as leaves age reduces physiological activity in a means of increasing nitrogen use efficiencies (Vitousek, 1982; Starr et al., 2000). Seasonal changes were most pronounced in the deciduous shrubs and forb, and least apparent in the evergreens. For the graminoids, because new leaves are being produced during much of the growing season (Shaver and Laundre, 1997), the seasonal patterns may not be a strong as those of the deciduous shrubs and forb. This seasonal pattern is partially responsible for the poor explanatory power of the regression analysis, which tests for linear relationships rather than the parabolic type response shown by seasonal photosynthetic capacity.
When comparisons were made among years, a decline in seasonal mean Amax in five of the study species was found during the 1999 field campaign compared with 1997–98. Declines were significant in four of the species, the two graminoids, V. vitis-idaea, and P. bistorta. The 1999 early season manipulation was 19 days as a consequence of unusually warm spring conditions and early snowmelt. Following the warm period, overall growing season temperatures were lower during 1999 compared with the two previous years, which may have led to lower overall Amax (Oberbauer et al., 1996). A week of near or below freezing daily minimum occurred in late July and is reflected in depressed physiological activity at that time. Reductions in stomatal conductances have been shown to occur following mid-growing season responses to freeze (Gorsuch and Oberbauer, 2002). Furthermore, an extended dry period occurred during the 1999 growing season. Although the growing season total (246 mm) was 23% more than in the 1998 field season, 34% of the total rainfall for 1999 occurred during two rain events (18 June and 17 July), thus significant dry periods between major rain events occurred. This lack of rain may have produced a moderate water stress that likely caused the decrease in Amax (Geiger and Sevaites, 1994), particularly in the more shallow-rooted evergreen shrubs and the forb (Kummerow and Russell, 1980). In contrast, the two deciduous species had higher Amax in 1999, with the increase statistically significant for S. pulchra. The difference appears to be a result of both species maintaining higher Amax later in the 1999 season than in the prior two years.
The results of this study provide evidence that photosynthetic capacity of key arctic species is relatively fixed over the course of the growing season, even in response to a substantial seasonal manipulation. These results have important implications for predicting the carbon balance of tundra ecosystems as the arctic climate changes. Modeling efforts may be simplified in that they can focus more on leaf area changes of species in response to climate change than on acclimations of area-based physiological activity. Given the large differences in photosynthetic capacity among species found in this and other studies, changes in vascular plant species composition will have large effects on ecosystem photosynthetic capacity. As temperatures in the Arctic have increased over the past two decades, tundra cryptogams have been declining (ACIA, 2005). This loss of cryptogams will increase the proportion of the total ecosystem photosynthetic production coming from vascular species, thereby increasing the effects of changes in photosynthesis, leaf area, and composition of the vascular species on total ecosystem photosynthesis.
Acknowledgments
This material is based in part on support by the National Science Foundation Office of Polar Programs grants OPP-9321626 and OPP-9615845 as part of the International Tundra Experiment Program (ITEX). Logistical support by Toolik Field Station staff and access to the LTER weather data by the Ecosystems Center of the Marine Biological Laboratory is greatly appreciated. Tracey Baldwin, Michael Rasser, Eric W. Pop, and Espy Rodriguez made invaluable contributions to the collection of the field data. Christina L. Staudhammer, Chad E. Husby, and Carlos A. Gonzalez assisted with the statistical analysis. We also thank two anonymous reviewers for their helpful comments.